For almost a year, the most prominent products of my company, Molecular Materials Informatics, have been available for Apple’s touchscreen devices, namely the iPhone, iPod and iPad trilogy. They can be purchased from the iTunes AppStore, and nowhere else. The properties of the AppStore, and the policies of its reviewers, have gotten some rather awful press over the last couple of years, and have become quite infamous in some social circles.
Month: April 2011
An inline abbreviation mechanism for structure diagrams
One of the habits that most chemists engage in when they sketch out structures with a pen and paper is the use of abbreviations for certain kinds of functional groups. Using abbreviations saves a lot of wasted time and ink, and it is also a useful way to draw attention to the chemistry that is relevant to the subject at hand, and away from the spinach that is coming along for the ride. For example:
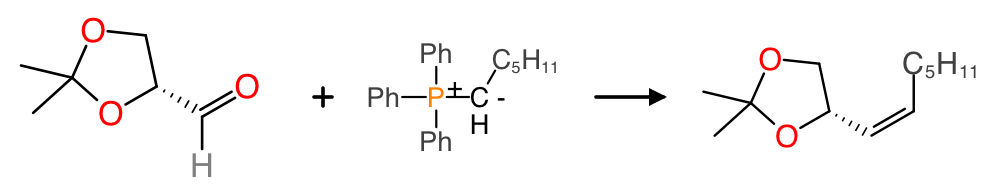 In this classical organic chemisty reaction, the phenyl groups of the Wittig reagent are abbreviated as Ph, and the unreactive alkyl chain is abbreviated as a molecular formula, because these groups are inert and not particularly exciting; the part that really matters is the reaction transform.
In this classical organic chemisty reaction, the phenyl groups of the Wittig reagent are abbreviated as Ph, and the unreactive alkyl chain is abbreviated as a molecular formula, because these groups are inert and not particularly exciting; the part that really matters is the reaction transform.
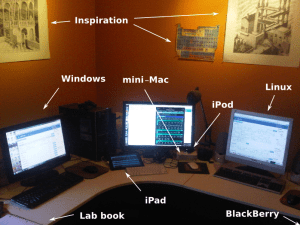
An ecumenical office space
One of the side-effects of developing software for a variety of different platforms is the need to use multiple desktop platforms for development and testing. My virtualisation platform is old school: a swivel chair.
All of the 3 major desktop platforms get equal facetime, although admittedly the Windows box is closest to the actual window. There may also be a PlayStation hiding in this picture somewhere (not labelled).
Excellent Mobile App Listing
Courtesy of Chris Swain, an excellent compilation of mobile apps can be found here:
http://homepage.mac.com/swain/Macinchem/iPhone/mobile_apps.html
The current menagerie: MMDS, Reaction101, Yield101
 As of the time of writing, the number of iOS apps published by Molecular Materials Informatics is about to grow to three, with the addition of Yield101, which is in the final stages of testing prior to submitting to the app store. All of these products are built around the same technology core, which includes, most importantly, a chemical structure diagram editor that is optimised for smartphone/tablet-sized devices. They also share a common visual theme and user interface style elements.
As of the time of writing, the number of iOS apps published by Molecular Materials Informatics is about to grow to three, with the addition of Yield101, which is in the final stages of testing prior to submitting to the app store. All of these products are built around the same technology core, which includes, most importantly, a chemical structure diagram editor that is optimised for smartphone/tablet-sized devices. They also share a common visual theme and user interface style elements.
The apps, in chronological order of release are… Continue reading
Why have chemistry software on mobile devices?
As the founder of Molecular Materials Informatics, and the programmer/scientist who designed and built its mobile chemistry products, a sensible question to address is: what is the point of going to all this trouble to redesign the core user interfaces of chemical information software so that it works well on things like phones and tablets?
To many of the people who already own mobile devices and enjoy using them in preference to more conventional computing hardware, this question probably answers itself, but to many others, smartphones and tablets are just tools that have some role to play. Why does having apps to create, manage and exchange chemical data make sense – and why would somebody think that was enough of a business opportunity to justify quitting a great job and starting a new company? Continue reading
Inaugural post
Welcome to the Cheminformatics 2.0 blog. The name is an obvious riff on the “web 2.0” moniker: the subject relates to the design and use of chemical information software on such new-fashioned platforms as mobile devices and feature-rich client-heavy web pages, as well as other related modern paradigms such as cloud-computing and distributed storage of data and computational capacity.
Blogs that will be appearing under this label will have a much lower density of trendy buzzwords than the preceding paragraph. Rather the articles will be oriented toward describing recent progress toward building new products, or finding new uses for them. The theme is extension of chemistry software to areas that go beyond the traditional domain of workstation class computers.