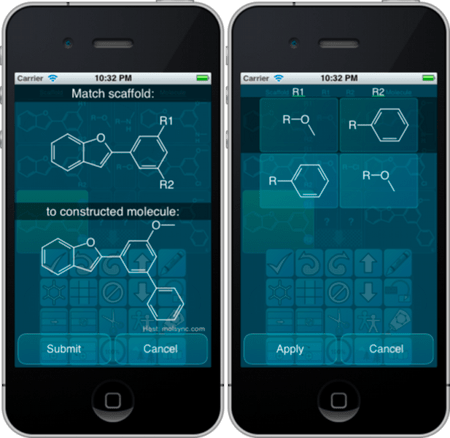
Version 1.1 of the SAR Table app just passed review and is available on the iTunes AppStore. This release is an important one, because it adds a major new feature: scaffold matching. For a row which has a whole molecule defined, and a scaffold core, the scaffold matching algorithm can be invoked to figure out the substituents:
The matching is done by outboarding to a remote procedure call, so the specialised substructure search is not done on the device itself. The actual functionality goes quite deep into the heart of cheminformatics, and is tailored for the specific needs of this app. For example, the scaffold can be pre-decorated by substituents, e.g. R1, R2, etc., or if not, they will be created automatically. Symmetry and other forms of degeneracy are handled in various ways. There is often more than one way to label a scaffold and assign the substituents, and ultimately the user is offered a choice, with various ranking and pruning activities handled behind the scenes.
The workflow implications of this new feature are important. The SAR Table app was designed first and foremost as a way to enter scaffold/substituent content, by re-using scaffold and substituent fragments and automatically building the construct molecule to keep it consistent with the component pieces. But there are quite a few possible workflow scenarios where the whole molecule is already available, e.g. if you have them in an SD file, it is rather handy to be able to import these structures into SAR Table, and then decompose them into fragments. If the scaffolds are known, or a quick guess can be made easily enough by picking representative structures and pruning them down, the series decomposition simply involves pasting the scaffold, activating the match feature, and approving the result.
It’s hard to explain the whole thing in a paragraph, but there is a much longer article that introduces the new functionality as a tutorial, with a lot of screenshots: see Scaffold Matching. There is also a Demo Video introducing this feature, which is probably a better place to start if you have a few minutes to spare.
The underlying algorithm that provides this feature is 100% original code, and is based on the same software stack that powers molsync.com. This is just the latest of many high-powered cheminformatics capabilities that will be made available to apps from Molecular Materials Informatics.